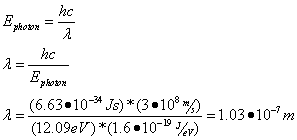
Tag Archives: photoelectrons
Photoelectric Effect / Models of the Atom
Models of the Atom
In the early 1900s, scientists around the world began to refine and revise our understanding of atomic structure and sub-atomic particles. Scientists understood that matter was made up of atoms, and J.J. Thompson had shown that atoms contained very small negative particles known as electrons, but beyond that, the atom remained a mystery.
Rutherford Model

New Zealand scientist Ernest Rutherford devised an experiment to better understand the rest of the atom. The experiment, known as Rutherford’s Gold Foil Experiment, involved shooting alpha particles (helium nuclei) at a very thin sheet of gold foil, and observing the deflection of the particles after passing through the gold foil. Rutherford found that although most of the particles went through undeflected, a significant number of alpha particles were deflected by large amounts. Using an analysis based around Coulomb’s Law and the conservation of momentum, Rutherford concluded that:
- Atoms have a small, massive, positive nucleus at the center.
- Electrons must orbit the nucleus.
- Most of the atom is made up of empty space.
Rutherford’s model was incomplete, though, in that it didn’t account for a number of effects predicted

by classical physics. Classical physics predicted that if the electron orbits the atom, it is constantly accelerating, and should therefore emit photons of EM radiation. Because the atom emits photons, it should be losing energy, therefore the orbit of the electron would quickly decay into the nucleus and the atom would be unstable. Further, elements were found to emit and absorb EM radiation only at specific frequencies, which did not correlate to Rutherford’s theory.
Bohr Model

Following Rutherford’s discovery, Danish physicist Niels Bohr traveled to England to join Rutherford’s research group and refine Rutherford’s model of the atom. Instead of focusing on all atoms, Bohr confined his research to developing a model of the simple hydrogen atom. Bohr’s model made the following assumptions:
- Electrons don’t lose energy as they accelerate around the nucleus. Instead, energy is quantized… electrons can only exist at specific discrete energy levels.
- Each atom allows only a limited number of specific orbits at each energy level.
- To change energy levels, an electron must absorb or emit a photon of energy exactly equal to the difference between the electron’s initial and final energy levels:
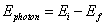
Question: Calculate the energy of the emitted photon when an electron moves from an energy level of -1.51 eV to -13.6 eV.
Answer:
Question: What is the emitted photon’s wavelength?
Answer:
Bohr’s Model, therefore, was able to explain the first two limitations of Rutherford’s Model. Further, Bohr was able to use his model to predict the frequencies of photons emitted and absorbed by hydrogen, explaining Rutherford’s problem of emission and absorption spectra! For his work, Bohr was awarded the Nobel Prize in Physics in 1922.
"If quantum mechanics hasn’t profoundly shocked you, you haven’t understood it yet." — Niels Bohr
Wave-Particle Duality
Modern Physics refers largely to advancements in physics from the 1900s to the present, extending the models of Newtonian (classical) mechanics and electricity and magnetism to the extremes of the very small, the very large, the very slow and the very fast. Modern Physics can encompass a tremendous variety of topics, which include:
- models of the atom
- sub-atomic structure
- universal conservation laws
- mass-energy equivalence
- fundamental forces in the universe
- the dual nature of electromagnetic radiation
- the quantum nature of atomic energy levels
Although electromagnetic waves exhibit many characteristics and properties of waves, they can also exhibit some characteristics and properties of particles. These “particles” are called photons. Light (and all EM radiation), therefore, has a dual nature. At times, light acts like a wave, and at other times it acts like a particle.
Characteristics of light that indicate light behaves like a wave include: Diffraction, Interference, Doppler Effect, and Young’s Double-Slit Experiment.
Characteristics of light that indicate light also acts as a particle include Blackbody Radiation, the Photoelectric Effect, and the Compton Effect.
“God does not play dice with the cosmos.” — Albert Einstein
“Einstein, don’t tell God what to do.” — Niels Bohr
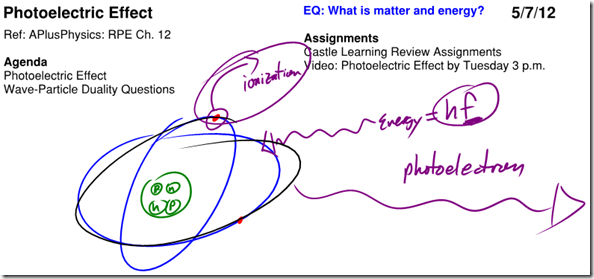
Photoelectric Effect
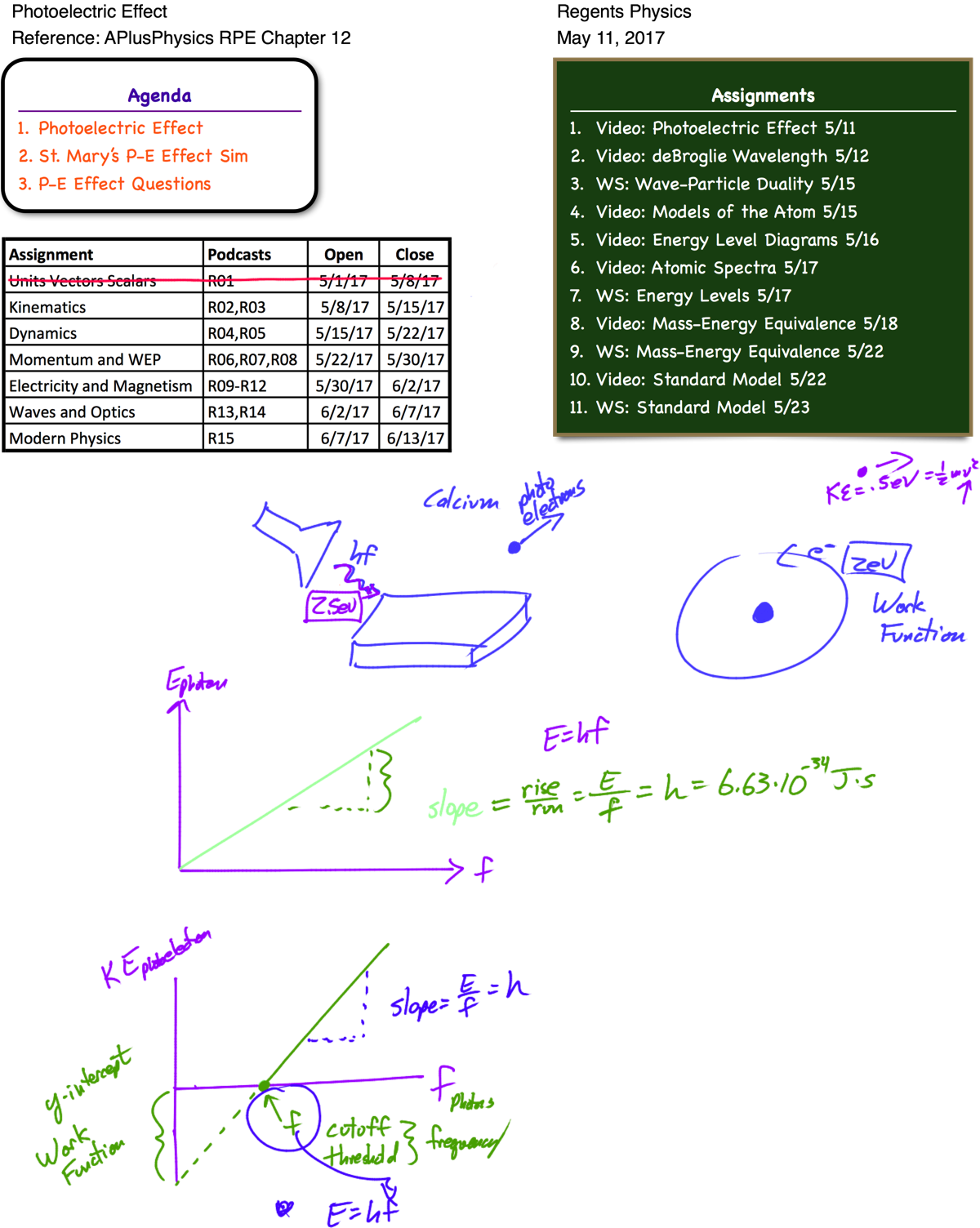
Further evidence that light behaves like a particle was proposed by Albert Einstein in 1905. Scientists had observed that when EM radiation struck a piece of metal, electrons could be emitted (known as photoelectrons). What was troubling was that not all EM radiation created photoelectrons. Regardless of what intensity of light was incident upon the metal, the only variable that effected the creation of photoelectrons was the frequency of the light. If energy exists only in specifi c, discrete amounts, EM radiation exists in specifi c discrete amounts, and these smallest possible “pieces” of EM radiation are known as photons. A photon has zero mass and zero charge, and because it is a type of EM radiation, its velocity in a vacuum is equal to c (3×108 m/s). The energy of each photon of light is therefore quantized and is related to its frequency by the equation:
![]()
In this equation, the value of h, known as Planck’s Constant, is given as 6.63×10-34 J•s. Einstein proposed that the electrons in the metal object were held in an “energy well,” and had to absorb at least enough energy to pull the electron out of the energy well in order to emit a photoelectron. The electrons in the metal would not be released unless they absorbed a single photon with that minimum amount of energy, known as the work function (φ) of the metal. The frequency of this photon is known as the cutoff frequency of the metal. Any excess absorbed energy beyond that required to free the electron became kinetic energy for the photoelectron.
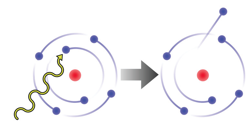
When a high-energy photon of light with energy greater than the energy holding an electron to its nucleus is absorbed by an atom, the electron is emitted as a photoelectron. The kinetic energy of the emitted photoelectron is exactly equal to the amount of energy holding the electron to the nucleus (the work function) subtracted from the energy of the absorbed photon.
![]()
This theory extended Planck’s work and inferred the particle-like behavior of photons of light. Photoelectrons would be ejected from the metal only if they absorbed a photon of light with frequency greater than or equal to a minimum threshold frequency, corresponding to the energy of a photon equal to the metal’s “electron well” energy for the most loosely held electrons. Regardless of the intensity of the incident EM radiation, only EM radiation at or above the threshold frequency could produce photoelectrons.