The current model of sub-atomic structure used to understand matter is known as the Standard Model. Development of this model began in the late 1960s, and has continued through today with contributions from many scientists across the world. The Standard Model explains the interactions of the strong (nuclear), electromagnetic, and weak forces, but has yet to account for the gravitational force. The search for the theorized Higgs Boson at Fermilab and CERN is an attempt to better unify and strengthen the Standard Model.
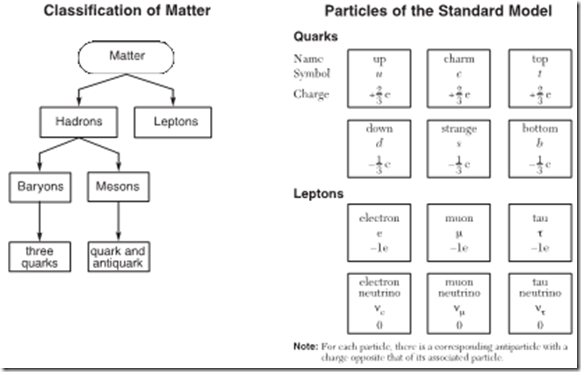
Although the Standard Model itself is a very complicated theory, the basic structure of the model is fairly straightforward. According to the model, all matter is divided into two categories, known as hadrons and the much smaller leptons. All of the fundamental forces act on hadrons, which include
particles such as protons and neutrons. In contrast, the strong nuclear force doesn’t act on leptons, so only three fundamental forces act on leptons such as electrons, positrons, muons, tau particles and neutrinos.
Hadrons are further divided into baryons and mesons. Baryons such as protons and neutrons are composed of three smaller particles known as quarks. Charges of baryons are always whole numbers. Mesons are composed of a quark and an anti-quark (for example, an up quark and an antidown
quark). If this sounds like a lot to keep track of, have no fear, this is summarized for you on the Regents Physics Reference Table.
Scientists have identified six types of quarks. For each of the six types of quarks, there also exists a corresponding anti-quark with an opposite charge. The quarks have rather interesting names: up quark, down quark, charm quark, strange quark, top quark, and bottom quark. Charges on each quark are either one third of an elementary charge, or two thirds of an elementary charge, positive or negative, and the quarks are symbolized by the first letter of their name. For the associated anti-quark, the symbol is the first letter of the anti-quark’s name, with a line over the name. For example, the symbol for the up quark is u. The symbol for the anti-up quark is ū.
Similarly, scientists have identified six types of leptons: the electron, the muon, the tau particle, and the electron neutrino, muon neutrino, and tau neutrino. Again, for each of these leptons there also exists an associated anti-lepton. The most familiar lepton, the electron, has a charge of -1e. Its anti-particle, the positron, has a charge of +1e.